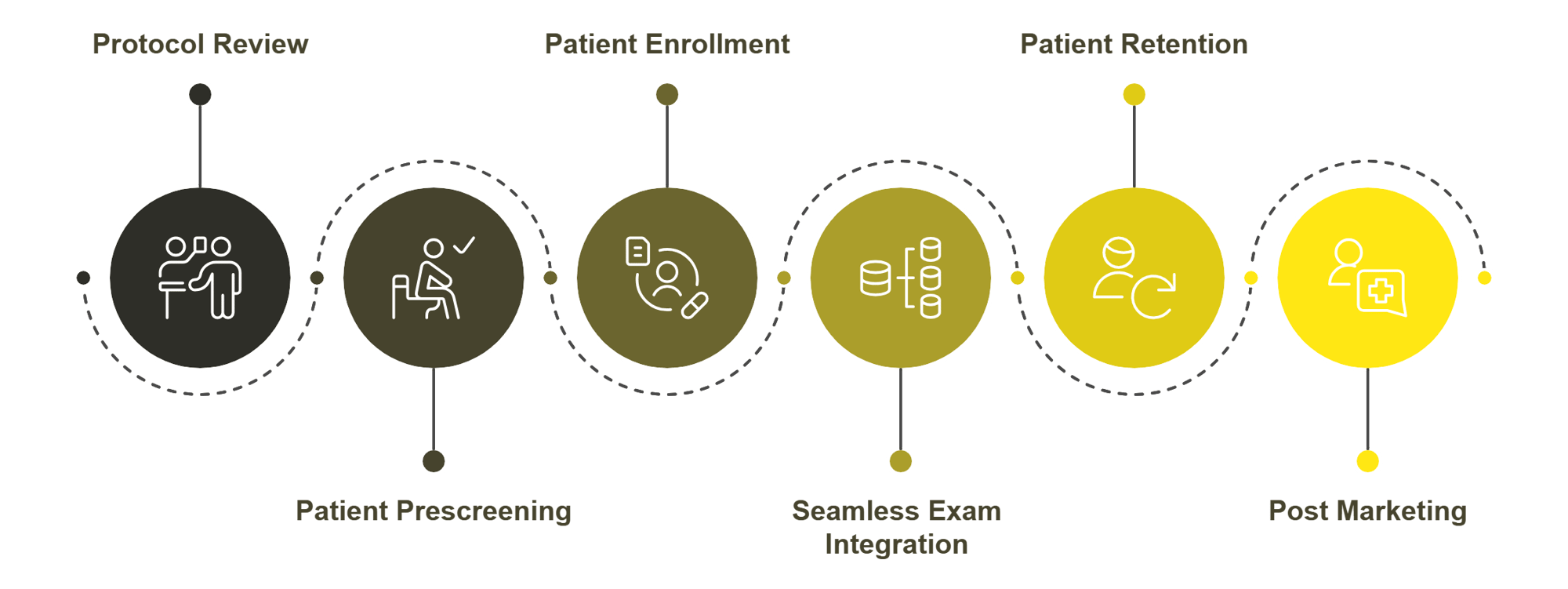
20/20 Onsite’s support for clinical trials stands apart from traditional, fixed-location sites due to its innovative approach, tailored to enhance participant accessibility, data quality, and trial efficiency. Here’s how 20/20 Onsite is different from legacy sites:
- Point-of-Need Accessibility: Unlike legacy sites, which require participants to travel, 20/20 Onsite brings state-of-the-art, point-of-need ophthalmic assessments directly to trial participants wherever they are—home, work, or even school.
- Patient-Centric Model: By minimizing patient travel and scheduling inconvenience, 20/20 Onsite increases engagement and retention. Our point-of-need clinics help ensure trials stay on track with high participant satisfaction and a 90+ Net Promoter Score.
- Enhanced Enrollment and Diversity: Fixed-location sites often limit access to patients within a specific geography, resulting in the underrepresentation of diverse populations. 20/20 Onsite’s point-of-need clinics enable recruitment from a broader and more diverse demographic, addressing a crucial need in clinical trials to meet enrollment goals effectively.
- Cost Control and Efficiency: Trial delays can cost sponsors millions daily. 20/20 Onsite's point-of-need service model is designed to meet strict timelines, reduce logistical delays, and enable sponsors to manage budgets better.
Overall, 20/20 Onsite offers a streamlined, patient-centered alternative that overcomes the traditional limitations of fixed-location clinical trial sites.





.png)